
Setting up mRNA manufacturing operations in other countries should start immediately, said Tom Frieden, the former director of the Centers for Disease Control and Prevention in the United States, adding: “They are our insurance policy against variants and production failure” and “absolutely can be produced in a variety of settings.” “You cannot go hire people who know how to make mRNA: Those people don’t exist,” the chief executive of Moderna, Stéphane Bancel, told analysts.īut public health experts in both rich and poor countries argue that expanding production to the regions most in need is not only possible, it is essential for safeguarding the world against dangerous variants of the virus and ending the pandemic. They say that the process is too complex, that it would be too time- and labor- intensive to establish facilities that could do it, and that they cannot spare the staff because of the urgent need to maximize production at their own network of facilities. Karan Deep Singh/The New York Timesĭespite mounting pressure, the chief executives of Moderna and Pfizer have declined to license their mRNA technology in developing countries, arguing it makes no sense to do so. Uttarakhand officials said the state will import 20 lakh doses of Sputnik vaccine over the next two months.Ĭumulatively, India's vaccination coverage stands at 17.72 Crores as of May 13 which is equivalent to 13.55% (approx) of India's 1.3 billion people.Īmid demands to ramp up domestic supply, Serum Institute and Bharat Biotech have submitted to the Centre their production plan for the next four months, informing that they can scale it up to 10 crore and 7.8 crore doses respectively by August, official sources said.Ĭurently, Bharat Biotech's Covaxin and Oxford-AstraZeneca's Covishield are the only two vaccines are being used in India's inoculation drive against the coronavirus.Workers inspecting and packing vials of the Oxford-AstraZeneca Covishield vaccine at a Serum Institute of India site in Pune, India. 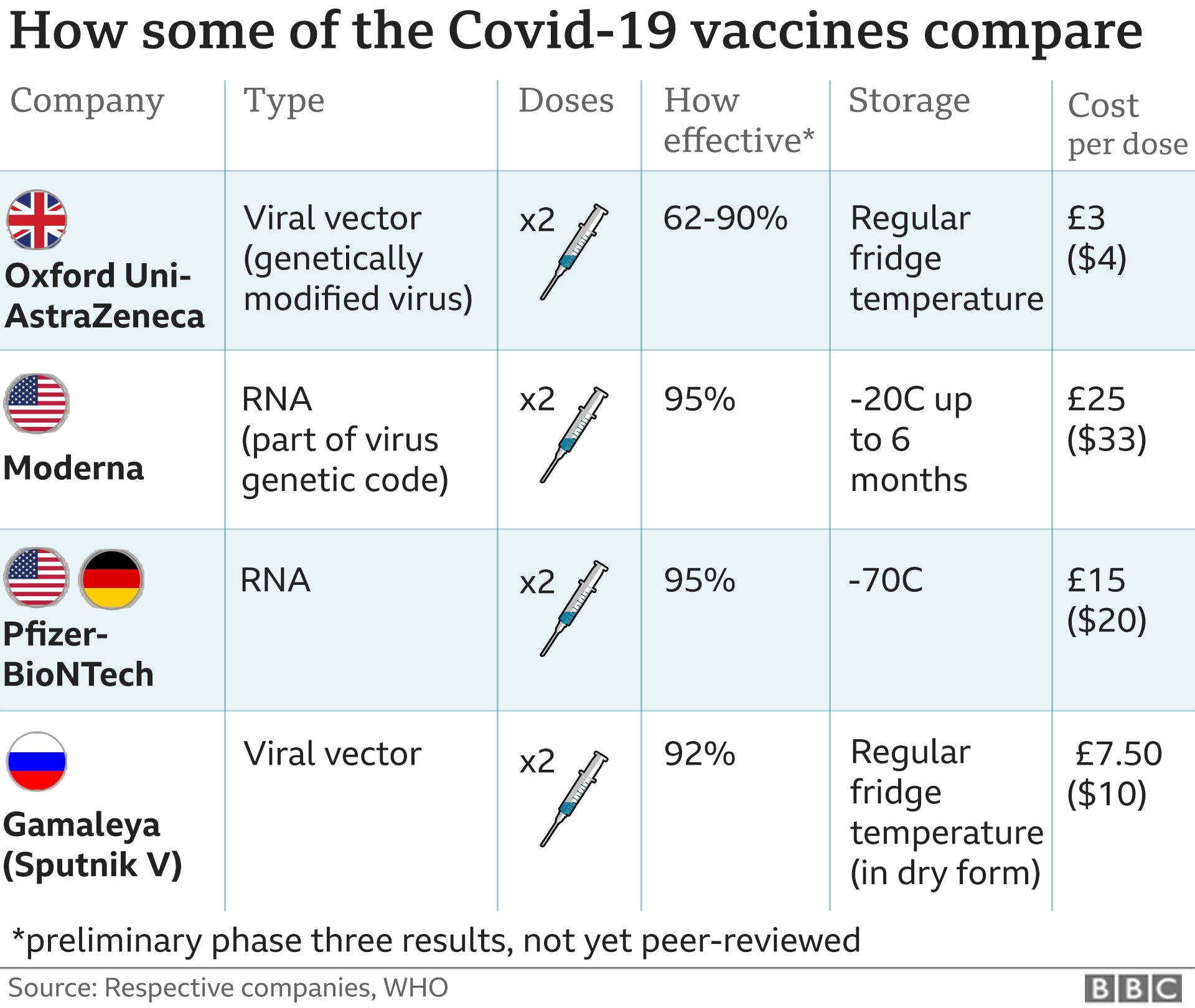
States like Tamil Nadu, Uttar Pradesh, Maharashtra, Karnataka, Andhra Pradesh, Telangana and Delhi have opted for global tenders to meet their needs. 
Besides, other states' governments have floated a tender to import vaccines. Several states in the country have suspended the coronavirus vaccination drive for the 18 to 44 age group as they have run out of stock. 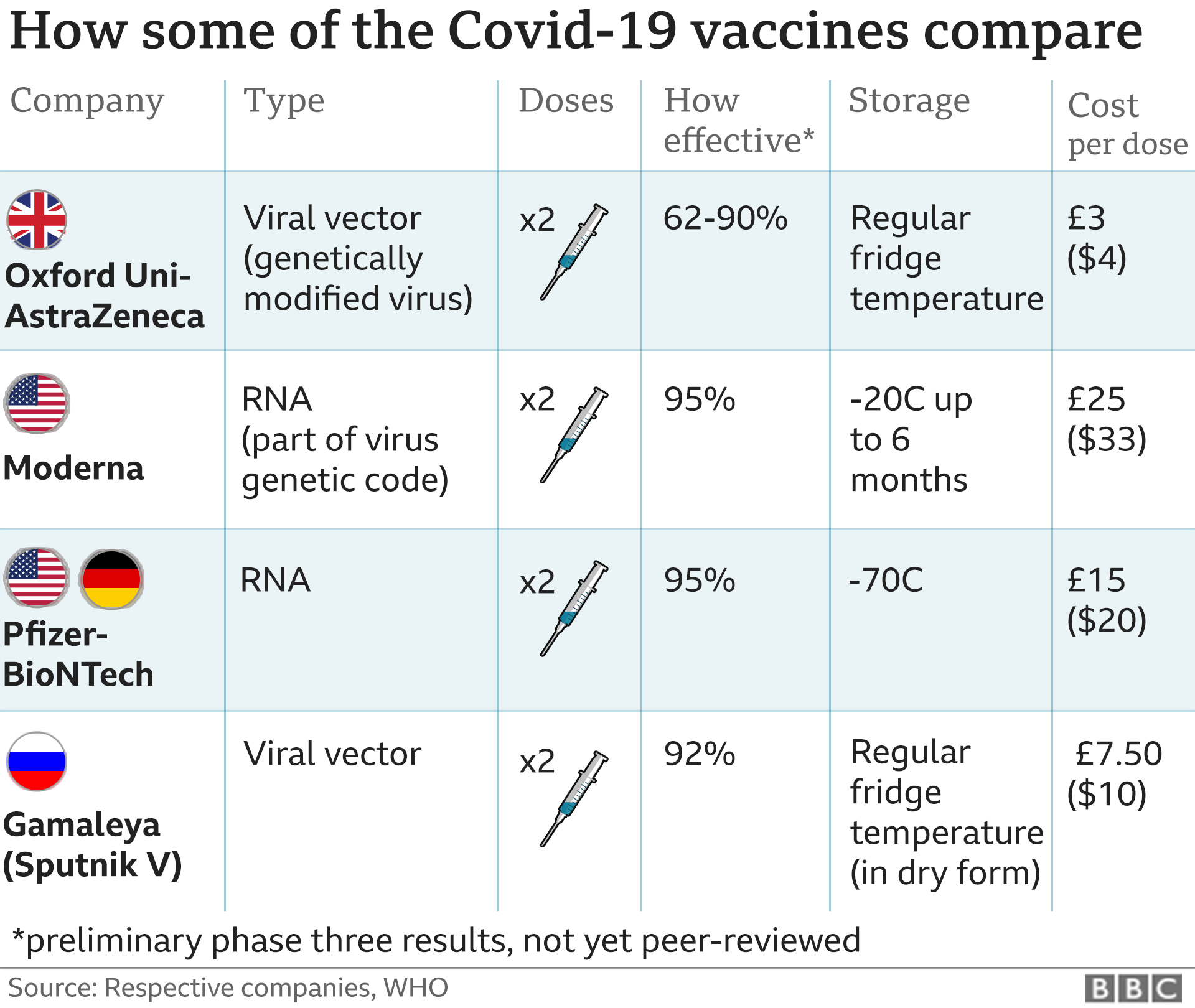
Meanwhile, Paul announced that Russia's Sputnik V will be available in markets across the country from early next week. The import license will be granted within 1-2 days". He added that "Any vaccine that is approved by FDA and the World Health Organization (WHO) can come to India. Yesterday, US Embassy said that the Biden administration is considering joint production of the Johnson & Johnson Covid-19 vaccine in India by tying up with like Serum Institute of India. Johnson & Johnson did a good job, they accepted this offer under Quad," Paul said on Thursday. "We invite them to manufacture here along with our companies. "I'm hopeful that they'll step forward to increase availability in India," he added.Īccording to Paul, among the three US-based coronavirus vaccine manufacturing companies, only Johnson & Johnson accepted Centre's offer. He added that the Government of India is connected with all three Covid-19 vaccine-making firms. Paul underscored that "They (Pfizer and Moderna) had said that they are working in their own way and they would talk of vaccine availability in Q3, in 2021".
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
